Illinois Bone & Joint Institute (IBJI), one of the largest independent, physician-owned orthopedic practices in the U.S., is one of the first in the state and the first in the Chicago suburbs outside of physicians who participated in the InSpace clinical trials, to use InSpace Balloon Implant technology.
InSpace is an advanced technology balloon implant to treat patients with massive irreparable rotator cuff tears (MIRCTs). Roger Chams, MD, an IBJI orthopedic surgeon with fellowship training in sports medicine and arthroscopy, performed his first InSpace balloon implantation on Friday, October 29.
Imagine a pillow that sits within the shoulder to support bone without anchors or permanent implant placement.
This innovative device has been shown to be superior to partial repair surgery, according to an AANA award-winning research paper that speaks to a randomized trial conducted by Nikhil N. Verma, MD. “The results of the study demonstrate the InSpace balloon is a ‘game-changer’ and presents a shorter, less invasive option that may enable sustained, clinically meaningful improvements in shoulder function and symptoms,” Dr. Verma stated in the study.
Dr. Roger Chams is one of four doctors in Illinois to have performed the procedure with multiple other patients scheduled.
“I’ve anticipated the FDA approval and release of this technology for years,” Dr. Chams says. “There is finally a good, safe, and minimally invasive alternative for patients with irreparable rotator cuff tears that have minimal arthritic changes that would previously have underwent a much more invasive procedure with high complication rates.”
Manufactured by Stryker and cleared by the FDA in July of 2021, the InSpace balloon implant has been used in over 29,000 procedures worldwide in over 40 countries. It offers an innovative solution to a complex problem with a shorter, minimally invasive procedure to improve mobility and reduce pain.
Step-by-Step Approach
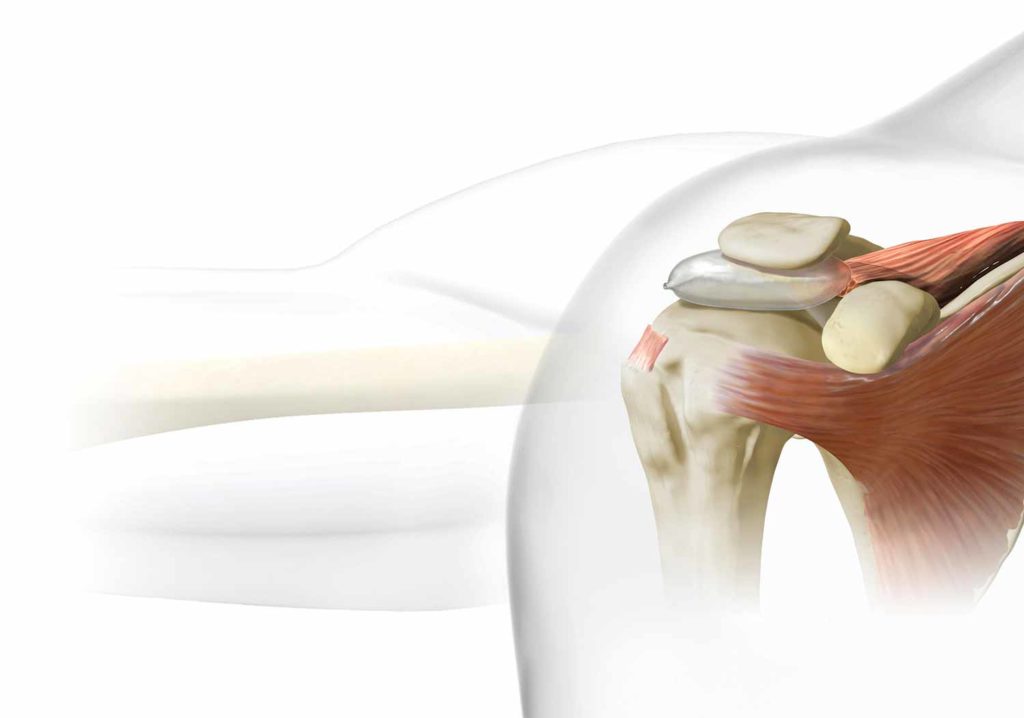
The InSpace Balloon Implant improves shoulder function and symptoms for patients with irreparable rotator cuff tears.
Two small incisions are made to allow room for the implant and camera to be introduced inside the shoulder cavity. A measurement is taken to select the appropriate sized implant to suit the patient’s anatomy. Next, the surgeon gently pushes the implant inserter into the shoulder cavity. The inserter has a deflated balloon at the end of it, and once it is positioned accurately using specified measurements, a saline solution inflates the balloon and the instruments are retracted. The balloon implant is designed to restore the subacromial space without sutures or fixation devices.
The average time for completing the procedure is 45 minutes, with the actual insertion of the balloon implant taking just a few minutes.
Who It’s For
Patients who underwent the InSpace balloon implant procedure previously suffered with pain and extremely limited mobility. They now have full motion and are able to lift weights, play tennis, sleep on their sides, and lift their grandchildren, all without pain.
*The InSpace™ subacromial tissue spacer system is indicated for the treatment of patients with massive, irreparable full-thickness torn rotator cuff tendons due to trauma or degradation with mild to moderate gleno-humeral osteoarthritis in patients greater than or equal to 65 years of age whose clinical conditions would benefit from treatment with a shorter surgical time compared to partial rotator cuff repair.
For more information on the InSpace balloon implant, visit stryker.com/inspace.




